Released March 2018
Introduction
This position statement has been developed to reflect the guidelines as set out by the National Health and Medical Research Council, Ethical Guidelines on the use of assisted reproductive technology in clinical practice and research (2017) (“NHMRC Ethical Guidelines”) and Fertility Solutions clinic policy.
Gamete refers to sperm and eggs whilst an embryo refers to an egg that has been successfully fertilized by a sperm.
Ethical Issues Of Gamete/Embryo Storage And Disposal
There are many complex ethical, religious and emotive issues associated with gamete/embryo storage, gamete/embryo donation and allowing gametes/embryos to succumb. The existence of diverse views on when life begins and the experience and outcomes of an individual/couples fertility treatment will impact on how participants approach end of storage decision-making.1
INDEFINITE STORAGE IS NOT AN OPTION.
It is a patients’ responsibility to inform Fertility Solutions of any changes to their contact details and that they respond promptly to the clinic notification in the event that they wish to use or donate any unused embryos.
When a patient decides to dispose or use their gametes/embryos all outstanding accounts must be first settled.
Extenuating Circumstances
From time to time teenagers and young adults are required to store gametes due to medical reasons that may affect their ongoing fertility i.e. cancer treatment. These circumstances are extenuating and the maximum storage time of 15 years may not apply. These situations will be dealt with upon an individual basis by written application of the patient to the clinic Quality Management Committee.
NHMRC Ethical Guidelines-Compliance
Separate to the ethical issues expressed above, bodies offering assisted reproductive procedures in Australia must be accredited by the Fertility Society of Australia, which requires compliance with the NHMRC Ethical Guidelines as part of their accreditation process. Fertility Solutions complies with the NHMRC Ethical Guidelines.
The NHMRC Ethical Guidelines deal with the issue of gamete and embryo storage and disposal.
Paragraph 7 of the NHMRC Ethical Guidelines state:
7.2 Assess the suitability of continued (long term) storage of gametes and embryos
Whilst it is not desirable to leave embryos in storage indefinitely these decisions are often very difficult to make. The NHMRC Ethical Guidelines state:
7.2.1 Clinics should have policies that guide the clinical determination for continued storage of gametes and embryos.
As such Fertility Solutions has adopted the following policy that supersedes the policy relating to the National Health and Medical Research Council, Ethical Guidelines on the use of assisted reproductive technology in clinical practice and research (2007) (“NHMRC Ethical Guidelines”).
Position Statement – Storage and Disposal of Gametes (Sperm and Eggs) and Embryos
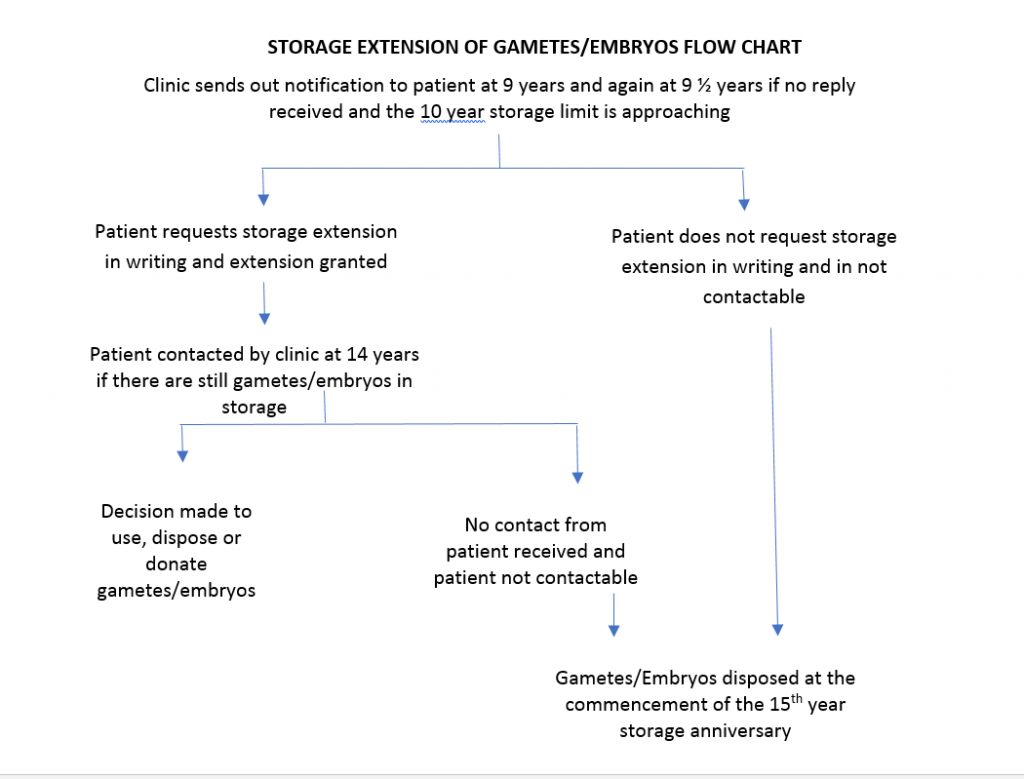
It is Fertility Solutions policy to provide gamete/embryo storage for 10 years with the option to renew consent for a further 5 years equating to a maximum of 15 years storage.
Reasons for 10+5 Years Storage
There are a number of reasons why it is Fertility Solutions policy to only store gametes/embryos for a maximum of 15 years. They are:
- To prevent the endless, expensive storage of abandoned gametes/embryos.2
- To prevent gametes/embryos from being held in storage after contact is lost with the 3 It is often exceedingly difficult to locate most of the individuals concerned and obtain up to date instructions about what to do with their gametes/embryos.
- To avoid the creation of a growing bank of frozen gametes/embryos.4
- To avoid the possibility of “gamete/embryo banking” which may occur, where gametes and embryos remain in storage for extended periods of time and ultimately are not required for use in
Options for Patients when the 10 Years for Storage is approaching
- In the case of the initial 10-year period, a further 5 years of storage can be requested in writing to the clinic manager.
- Stored gametes/embryos can be used for patients own
- Patients may consent to have the gametes/embryos being removed from storage and
- Patients may consent for the gametes/embryos being donated for the use of infertility treatment of others. This can only occur if the embryos have not been created using donor sperm or eggs.
Options for when the 15 Year Maximum Period of Storage is approaching
Patients can elect to:
- Use the gametes /embryos for their own
- Consent to having all the remaining gametes/embryos removed from storage and respectfully allowed to
- Consent for all the remaining gametes/embryos to be donated for the use of infertility treatment of others. This can only occur if the embryos have not been created using donor sperm or eggs.
Our Procedures
When unused gametes/embryos are in storage at Fertility Solutions the following will occur:
- Invoices are routinely emailed to the patient(s) (unless requested otherwise), 6 monthly (minimum of 2 weeks before payment is due) reminding them that they have gametes stored at the clinic and that payment is due. With the invoice the following information is also provided:
- Embryo-Gamete Frozen Storage General Information options
- Nine (9) years after the gametes/embryos are cryopreserved (this includes gametes that are imported to Fertility Solutions from another clinic and reach the age of 9 years since created), Fertility Solutions will write to the patient(s) via post (unless requested otherwise) and remind them that there are still gametes/embryos in storage, advising them of their options and asking them to take action in accordance with the option chosen.
If a written response is not received within 6 months a reminder letter will be sent via post.
If advice has not been received in writing regarding a choice on or before the 15th anniversary of the storage of the gametes/embryos, the clinic will arrange for the disposal of the gametes/embryos by respectfully allowing them to succumb.
Please note any outstanding accounts will still be applicable after disposal occurs. If Fertility Solutions Staff cannot contact the patient, their account will be sent to the debt collection agency to purse. Patients will be liable for the debt collection fee.
1 Reproductive Technology Council ofWestern Australia, Policyon Embryos Storage and Applications to Extend Storage Beyond TenYears, Reproductive Technology Council, February 2010, 3.
2 Notebook/Talk of the Streets, London: Embryo Ethics, TIME INT’L, August 5, 1996, at 6.
3 The storage of human embryos, Karin J Dawson, IVF Unit, Institute of Obstetrics and Gynaecology, Royal, Postgraduate Medical School, Hammersmith Hospital, Du Cane Road, LondonW12 ONN, UK, Human Reproduction vol.12 no.1 pp.3-11, 1997
4 Saunders DM, Bowman MC, Greierson A, Garner F. Frozen embryos: too cold to touch? The dilemma ten years on. Hum Reprod 1995, 10: 3081-3085